OBJECTIVE: To learn about ionic and covalent bonds and explain how they are formed. To learn about the polar covalent bond.
What is a chemical bond? Although there are several possible ways to answer this question, we will define a bond as a force that holds groups of two or more atoms together and makes them function as a unit. For example, in water the fundamental unit is the HOOOH molecule, which we describe as being held together by the two OOH bonds. We can obtain information about the strength of a bond by measuring the energy required to break the bond, the bond energy.
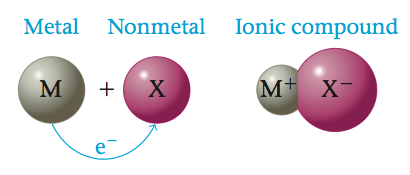
Ionic substances are formed when an atom that loses electrons relatively easily reacts with an atom that has a high affinity for electrons. In other words, an ionic compound results when a metal reacts with a nonmetal.
Ionic substances are formed when an atom that loses electrons relatively easily reacts with an atom that has a high affinity for electrons. In other words, an ionic compound results when a metal reacts with a nonmetal.
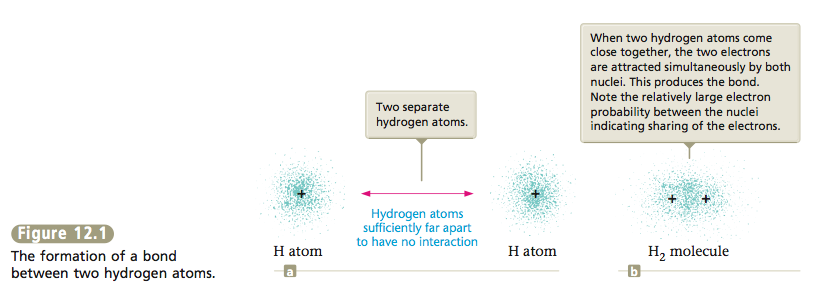
The type of bonding we encounter in the hydrogen molecule and in many other molecules where electrons are shared by nuclei is called covalent bonding.
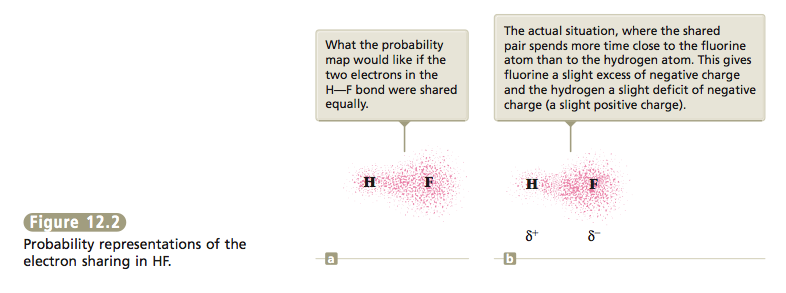
So far we have considered two extreme types of bonding. In ionic bonding, the participating atoms are so different that one or more electrons are transferred to form oppositely charged ions. The bonding results from the at- tractions among these ions. In covalent bonding, two identical atoms share electrons equally. The bonding results from the mutual attraction of the two nuclei for the shared electrons. Between these extremes are intermediate cases in which the atoms are not so different that electrons are completely transferred but are different enough so that unequal sharing of electrons results, forming what is called a polar covalent bond.